 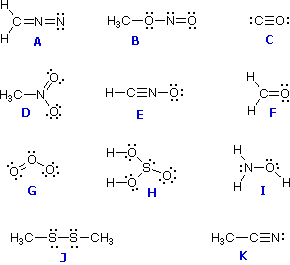
So three bonds, and withĪ formal charge of zero we know there should beĪ lone pair of electrons on that nitrogen. So here's one bond, twoīonds, and then three bonds. So we'll start with theĮxample on the left here and if we look at this nitrogen and we know it has aįormal charge of zero, let's see how many bonds it has. Let's look at otherĮxamples where nitrogen has a formal charge of zero. Leaving off the lone pair, because you should know it's there. Well if I just draw this and you know the formalĬharge of nitrogen is zero, then it's assumed you also know there's a lone pair of electrons on that nitrogen. And sometimes you don't want to draw in lone pairs of electrons, so So when nitrogen has three bonds and one lone pair of electrons, the formalĬharge is equal to zero. Lone pair of electrons, the formal charge is equal to zero. Nitrogen with three bonds, let me draw these in here, one, two, three. Here with our two hydrogens and a lone pair ofĮlectrons on the nitrogen. So in our drawing, nitrogen is surrounded by five valance electrons. Of electrons on the nitrogen, so that's four, and five. This would be one, two, three, and then we have a lone pair We have around nitrogen in our drawing? Let's count them up. Nitrogen gets one of the electrons and hydrogen gets the other. And same for this nitrogen-hydrogen bond. And from this bond on the right nitrogen gets one of those electrons and hydrogen gets the other. We know that from thisīond here on the left nitrogen gets one of those electrons. Here to the dot structure and let's look at these bonds. And from that we subtract the number of valence electrons that We know that nitrogen is supposed to have five valence electrons,īecause of its position on the periodic table. So formal charge is equal to the number of valence electrons that So I'm gonna draw in theĮlectrons in this bond so it's easier for us toĪssign a formal charge to the nitrogen. So let's assign a formalĬharge to the nitrogen in this molecule. Or another way of saying that, formal charge is equal to the number of valence electrons theĪtom is supposed to have minus the number of valence electrons that the atom actually has in the drawing. Number of valence electrons in the bonded atom. Number of valance electrons in the free atom minus the Write the Lewis structure and chemical formula of the compound with a molar mass of about 70 g/mol that contains 19.7% nitrogen and 80.We'll assign formal charge to nitrogen, and just to remind you of the definition for formal charge, formal charge is equal to the.Write Lewis structures for each of the four compounds and determine the formal charge of the iodine atom in each molecule: Iodine forms a series of fluorides (listed below).Is the actual structure consistent with the formal charges? Draw the structure of hydroxylamine, H 3NO, and assign formal charges look up the structure.Based on formal charge considerations, which of the following would likely be the correct arrangement of atoms in sulfur dioxide: OSO or SOO?.Based on formal charge considerations, which of the following would likely be the correct arrangement of atoms in hypochlorous acid: HOCl or OClH?.Based on formal charge considerations, which of the following would likely be the correct arrangement of atoms in nitrosyl chloride: ClNO or ClON?. 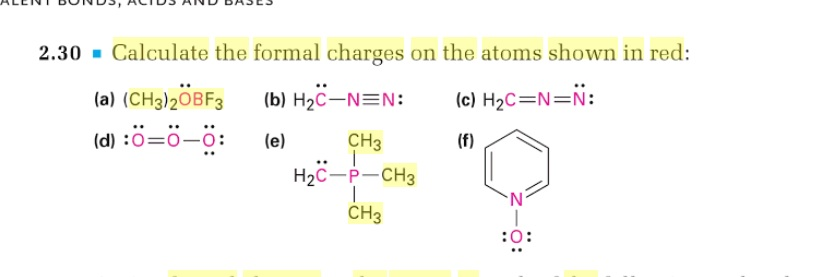
Determine the formal charge on each atom in each of the resonance structures:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
